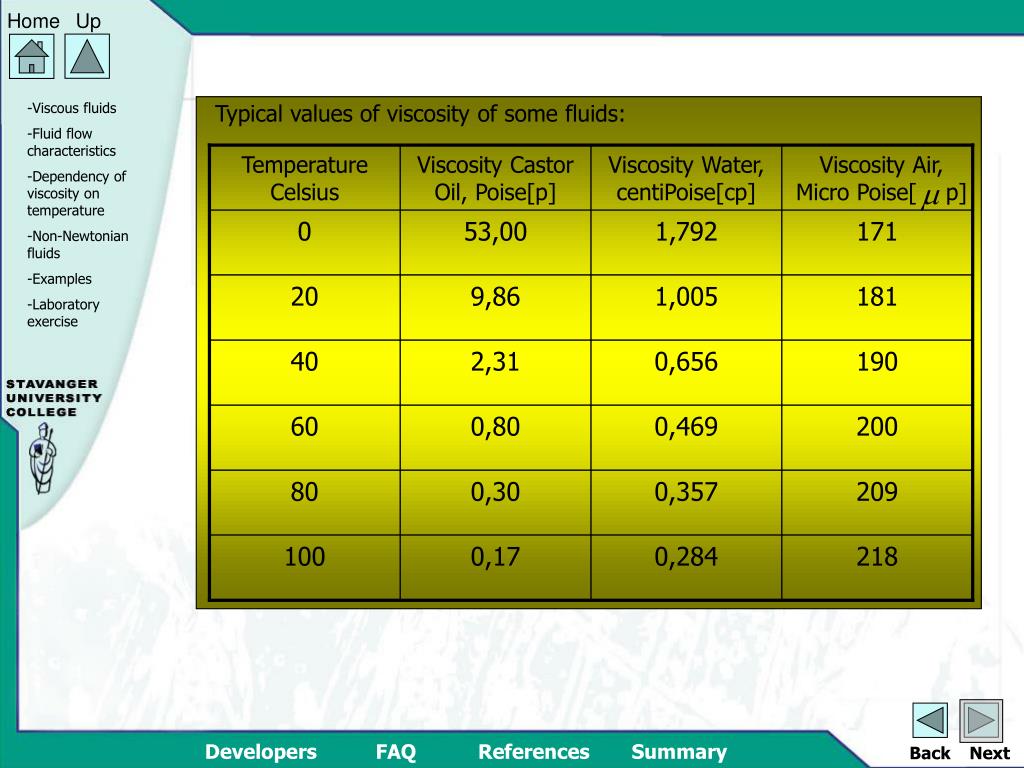 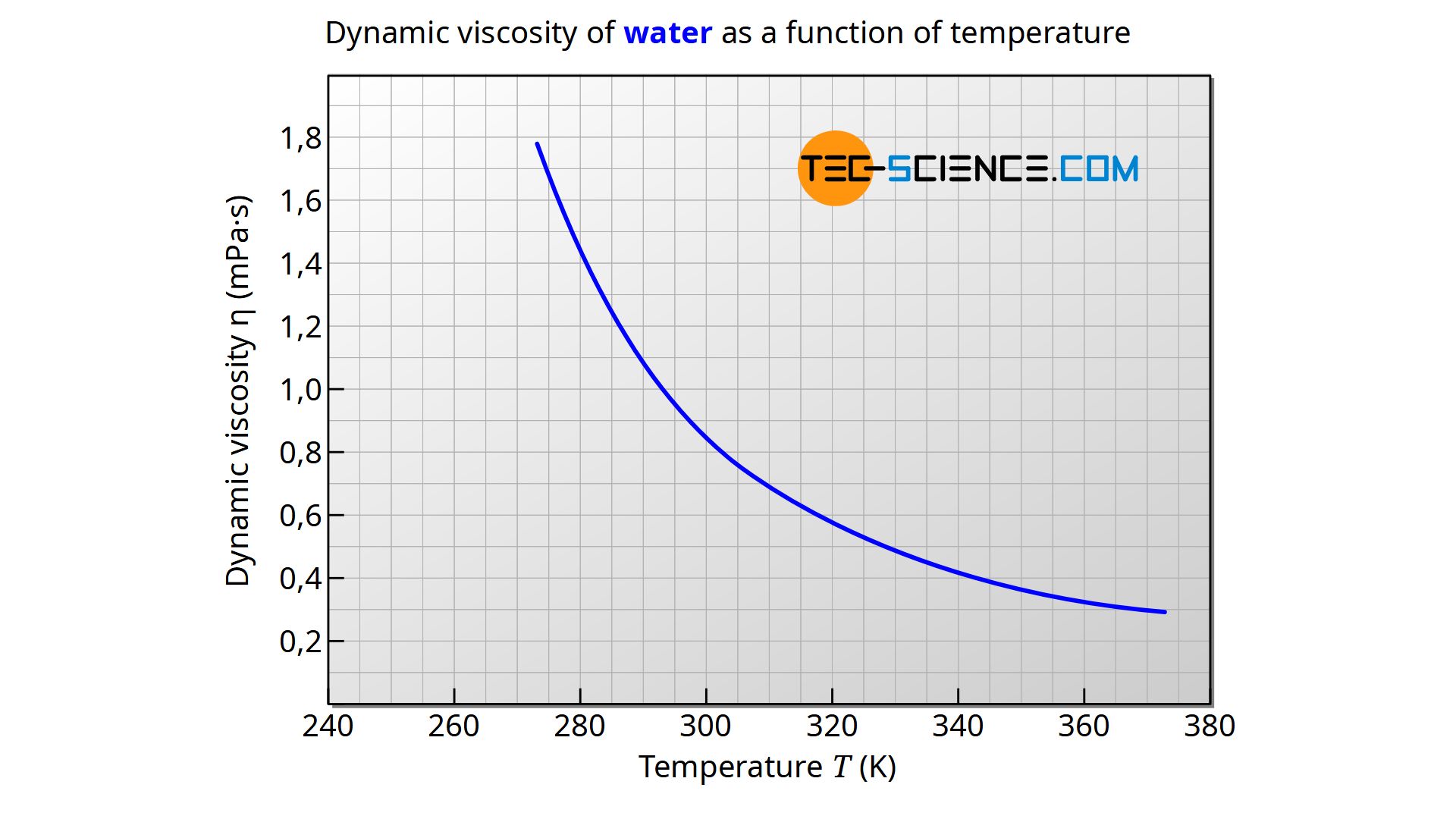
Kinematic viscosity of oxygen at gas-liquid equilibrium pressure:ĭynamic (absolute) and kinematic viscosity of oxygen at given temperatures and pressures, SI and Imperial units:įor full table with Dynamic and Kinematic Viscosity - rotate the screen! Oxygen - Dynamic and Kinematic Viscosity vs. Oxygen dynamic and kinematic viscosity at atmospheric pressure and varying temperature:ĭynamic viscosity of oxygen at varying temperature and 1, 10, 50 and 100 bara (14.5, 145, 7 psia):ĭynamic viscosity of oxygen at gas-liquid equilibrium pressure: See also other properties of Oxygen at varying temperature and pressure: Density and specific weight and Specific heat (heat capacity), and Thermophysical properties at standard conditions,Īs well as dynamic and kinematic viscosity of air, ammonia, benzene, butane, carbon dioxide, ethane, ethanol, ethylene, methane, methanol, nitrogen, propane and water. While the kinematic viscosity is given as cSt, m 2/s, and ft 2/s The output dynamic viscosity is given as Pa*s, N*s/m 2, cP, mPa*s, lb f*s/ft 2 and lb m/(ft*h), The calculator below can be used to estimate oxygen dynamic or kinematic viscosity at given temperatures and atmospheric pressure. Oxygen phase diagram Online Oxygen Viscosity Calculator Water has a viscosity of 0.0091 poise at 25 C, or 1. Tabulated values and viscosity units conversion are given below the figures. The dynamic viscosity of water is 8.90 × 104 Pas or 8.90 × 103 dyns/cm2 or 0.890 cP at about 25 C. Absolute or dynamic viscosity is used to calculate Reynold's Number to determine if a fluid flow is laminar, transient or turbulent. Note Viscosity conversions are based on fluids with specific gravity of one. The viscosity of a fluid is a measure of its resistance to gradual deformation by shear stress or tensile stress.įor further definitions, go to Absolute (dynamic) and kinematic viscosity. 2) Water at 68.4oF (20.2oC) has an absolute viscosity of one - 1 - centiPoise.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
